About the Author
David Chen is a Clinical Engineering Consultant with 18 years of experience in healthcare technology management. He has served as Director of Clinical Engineering at two major US health systems—Memorial Hermann Health System (Houston, TX) and UCHealth (Aurora, CO)—where he led device standardization programs covering more than 5,000 clinical mobile devices across 25 hospitals. David holds a Master of Science in Biomedical Engineering from the University of Michigan and is a Certified Healthcare Technology Manager (CHTM) through AAMI. He has testified as an expert witness on medical device safety standards and has published peer-reviewed research on mobile device infection control in the Journal of Clinical Engineering. This guide draws on his direct experience evaluating, procuring, and deploying both consumer and medical-grade tablets in high-acuity clinical environments.
Introduction: The Tablet Dilemma in Modern Healthcare
A nurse walks into a patient room with an iPad. It looks familiar, feels intuitive, and costs a fraction of the dedicated clinical device sitting unused at the nursing station. The question echoes through hospital IT departments, clinical engineering meetings, and procurement offices across the healthcare industry: Why can’t we just use consumer tablets?
The answer lies not in capability but in compliance. The difference between consumer tablets and medical-grade terminals extends far beyond price tags and processor speeds. It encompasses regulatory requirements, patient safety standards, infection control protocols, clinical workflow integration, and total cost of ownership that, when fully accounted for, often reveals consumer devices as the more expensive option.
For hospital IT decision-makers, clinical engineering leaders, procurement specialists, and nursing administrators, understanding this distinction is essential. Deploying consumer tablets in clinical environments exposes organizations to regulatory risk, creates workflow friction, increases infection transmission vectors, and ultimately delivers a false economy.
This guide provides a comprehensive comparison of consumer vs medical grade tablets, examining the technical, regulatory, and operational dimensions that determine the right tool for clinical environments. Drawing on industry standards including IEC 60601, FDA guidance, and real-world deployment experience, it offers a framework for making informed mobile device investments that balance cost, compliance, and clinical utility.

1. Defining the Categories
1.1 Consumer Tablets in Healthcare
Consumer tablets—Apple iPad, Samsung Galaxy Tab, Amazon Fire, and similar devices—are designed for general-purpose use in home and office environments. They excel in user experience, application ecosystems, and affordability. However, they are not engineered for the unique demands of healthcare environments.
When deployed in clinical settings, consumer tablets face:
- Frequent failure from cleaning chemical exposure
- Battery degradation from continuous charging cycles
- Physical damage from drops and impacts
- Limited integration with clinical systems
- Absence of clinical-grade security features
1.2 Medical-Grade Terminals
Medical-grade tablets—also referred to as healthcare grade tablets or clinical tablets—are purpose-engineered for healthcare environments. These medical-grade mobile devices incorporate design elements, materials, and certifications that address the specific requirements of clinical use:
- Regulatory compliance with medical device standards
- Biocompatible materials that resist microbial colonization
- Clinical workflow integration with EHR and nurse call systems
- Enhanced durability for hospital environments
- Specialized power management for continuous operation
Unlike consumer devices repurposed for healthcare, medical-grade terminals are designed from the ground up for the clinical environment.
2. Compliance and Regulatory Standards
2.1 IEC 60601: The Medical Electrical Equipment Standard
The most significant distinction between consumer and medical-grade tablets is compliance with IEC 60601, the international standard for medical electrical equipment safety and performance.
IEC 60601 certification requires:
- Electrical safety: Leakage current limits that prevent harm to patients connected to other medical devices
- Electromagnetic compatibility: Immunity from interference with other medical equipment and assurance that the device does not cause interference
- Mechanical safety: Protection against moving parts, sharp edges, and other physical hazards
- Environmental testing: Operation under expected clinical conditions
A consumer tablet lacks IEC 60601 certification. When placed within six feet of a patient—the standard definition of the “patient environment”—these devices become potential sources of electrical hazard and electromagnetic interference with life-sustaining equipment such as ventilators, infusion pumps, and cardiac monitors.
2.2 FDA Classification
Some medical-grade tablets are classified as FDA Class I medical device. This classification, while representing the lowest risk category, indicates that the device has undergone regulatory review and is intended for use in patient care. Consumer tablets carry no such classification and are not recognized as medical devices by the FDA.
2.3 HIPAA Compliance and Security
HIPAA compliance requirements for devices handling protected health information (PHI) include:
- Encryption: Data must be encrypted at rest and in transit
- Access controls: Strong authentication mechanisms, typically single sign-on (SSO) integrated with clinical identity management systems
- Audit trails: Comprehensive logging of access to PHI
- Remote wipe: Ability to securely erase devices if lost or stolen
- Secure boot: Protection against unauthorized operating system modifications
While consumer tablets can be configured with some security features, medical-grade terminals ship with these capabilities built into the hardware and operating system, with clinical-grade management tools designed for healthcare IT environments.
2.4 UL 60601 and Other Certifications
Additional certifications relevant to healthcare deployments include:
- UL 60601: Underwriters Laboratories certification for medical electrical equipment
- RoHS: Restriction of hazardous substances in electronic equipment
- REACH: Registration, evaluation, authorization of chemicals in European markets
- TAA: Trade Agreements Act compliance for US government procurement
3. Infection Control: A Critical Differentiator
3.1 Antimicrobial Housing
Hospital-acquired infections (HAIs) affect hundreds of thousands of patients annually. Devices placed in patient rooms must not become vectors for pathogen transmission.
Medical-grade tablets incorporate antimicrobial housing materials:
- Silver-ion impregnated plastics that inhibit bacterial growth
- Copper alloy surfaces with natural antimicrobial properties
- Biocide additives integrated into the casing material
Consumer tablets use standard plastics that provide no inherent antimicrobial protection and may actually support bacterial colonization on porous surfaces.
3.2 Chemical Disinfection Compatibility
Healthcare environmental services teams use powerful disinfectants to clean patient rooms:
- Quaternary ammonium compounds (quats)
- Hydrogen peroxide-based cleaners
- Bleach solutions (sodium hypochlorite)
Medical-grade tablets are engineered with zero-pore surfaces that withstand thousands of cleaning cycles with these aggressive chemicals. Consumer tablets, by contrast, suffer material degradation from repeated chemical exposure. Screens develop micro-cracks, coatings peel, and ports corrode.
3.3 UV-C Disinfection Compatibility
Many healthcare facilities now use UV-C disinfection systems for terminal cleaning. UV-C disinfection compatible medical tablets can be safely exposed to germicidal ultraviolet light without damage to displays, sensors, or internal components. Consumer tablets are not rated for UV-C exposure and may suffer display degradation, plastic embrittlement, or seal failure.
3.4 Sealed Enclosures
Medical-grade tablets feature fully sealed enclosures that prevent fluid ingress. IP rating (Ingress Protection) levels for clinical devices typically range from IP54 to IP67, ensuring protection against dust and liquid exposure during cleaning and clinical use. Consumer tablets offer minimal ingress protection, leaving internal components vulnerable to damage from spills, cleaning solutions, or bodily fluids.
4. Durability and Reliability
4.1 MIL-STD-810 Testing
MIL-STD-810 testing, originally developed for military equipment, evaluates device durability against environmental stressors. Medical-grade tablets commonly undergo:
- Drop testing: Repeated drops onto hard surfaces from clinical use heights
- Vibration testing: Simulated transport and cart movement
- Temperature extremes: Operation in varied clinical environments
- Humidity resistance: Performance in high-moisture settings
Consumer tablets are not tested to these standards. A consumer tablet dropped on a hospital floor often results in a cracked screen, damaged display, or non-functional device. Medical-grade tablets withstand drops and continue operating.
4.2 Mean Time Between Failures
Medical-grade tablet durability testing standards translate into measurable reliability metrics. Medical-grade devices typically specify MTBF (Mean Time Between Failures) of 30,000 to 50,000 hours—years of continuous operation. Consumer tablets lack published MTBF data for clinical environments and typically fail within 12 to 18 months of deployment.
4.3 Warranty and Support
Medical-grade tablet warranty and support differences are substantial:
- Medical devices typically include 3 to 5 year warranties with advanced replacement options
- Support teams are trained in clinical environments and understand healthcare workflows
- Replacement devices are available with expedited shipping for clinical continuity
Consumer tablet warranties are designed for home use, typically 1 year with standard support. Replacements require shipping the device for repair, leaving clinical workflows disrupted for days or weeks.
5. Clinical Workflow Integration
5.1 Electronic Health Record Integration
The electronic health record (EHR) is the central nervous system of modern healthcare. Medical-grade tablets offer native integration with major EHR platforms including Epic, Cerner, and Meditech through:
- HL7 messaging protocols for clinical data exchange
- FHIR (Fast Healthcare Interoperability Resources) APIs for modern integration
- Pre-certified compatibility that eliminates custom development costs
EHR integration requirements for hospital tablets often include bidirectional data flow: patient identification, care team display, medication administration recording, and clinical documentation. Consumer tablets require middleware or custom application development to achieve similar integration, adding complexity and cost.
5.2 Nurse Call System Integration
Nurse call system integration enables patients to request assistance through the tablet interface. Medical-grade terminals connect directly to existing nurse call infrastructure, allowing:
- Patient-initiated requests with specific need categorization
- Request tracking and response time monitoring
- Cancellation capability to reduce unnecessary staff interruptions
Consumer tablets cannot integrate directly with nurse call systems without additional hardware and custom software development.
5.3 Point-of-Care Documentation
Point-of-care documentation—charting at the bedside rather than at a stationary workstation—improves accuracy and nursing efficiency. Medical-grade tablets support:
- Barcode scanner integration for medication administration verification
- Peripheral device connectivity for vital signs capture
- Fast boot and resume for immediate access between patient encounters
Clinical workflow efficiency gains from medical-grade tablets typically deliver 20 to 30 minutes per nurse per shift in reclaimed time.
5.4 Authentication and Access
Medical-grade tablets incorporate single sign-on (SSO) capabilities that integrate with hospital identity management systems. Clinicians tap their badge or use biometric authentication to access patient information without repeated password entry. Consumer tablets lack these authentication capabilities, leading to workarounds that compromise security or consume nursing time.
6. Power Management and Battery Life
6.1 Hot-Swappable Batteries
Hot-swappable battery capability allows clinicians to replace a depleted battery without powering down the device or interrupting the clinical session. A nurse mid-documentation can swap batteries and continue without rebooting.
Medical-grade tablets commonly feature hot-swappable batteries with:
- 8 to 12 hours of continuous operation per battery
- Smart charging in docking stations that prioritize battery health
- Battery status indicators visible at a glance
Consumer tablets have sealed batteries that cannot be replaced in the field, requiring devices to be taken out of service for charging or battery service.
6.2 Charging Infrastructure
Medical-grade tablets use purpose-designed docking stations that:
- Charge devices rapidly for quick turnaround between shifts
- Provide wired network connectivity for data synchronization
- Mount securely in clinical environments
- Support shared device models where tablets are assigned by shift
Consumer tablets require aftermarket charging carts and stands that lack clinical-grade durability and integration.
6.3 Battery Lifespan
Consumer tablet batteries typically endure 500 to 1,000 charge cycles before significant capacity degradation—approximately 18 to 24 months of daily use. Medical-grade tablets use higher-grade battery cells designed for 2,000 to 3,000 cycles, providing 4 to 5 years of service before battery replacement becomes necessary.
7. Total Cost of Ownership Analysis
7.1 Acquisition Cost Comparison
| Device Type | Typical Unit Cost | Quantity (100 devices) |
|---|---|---|
| Consumer Tablet (iPad/Samsung) | $500 – $1,000 | $50,000 – $100,000 |
| Medical-Grade Tablet | $1,500 – $3,500 | $150,000 – $350,000 |
The acquisition cost difference is substantial—medical-grade tablets typically cost 2 to 4 times more than consumer alternatives.
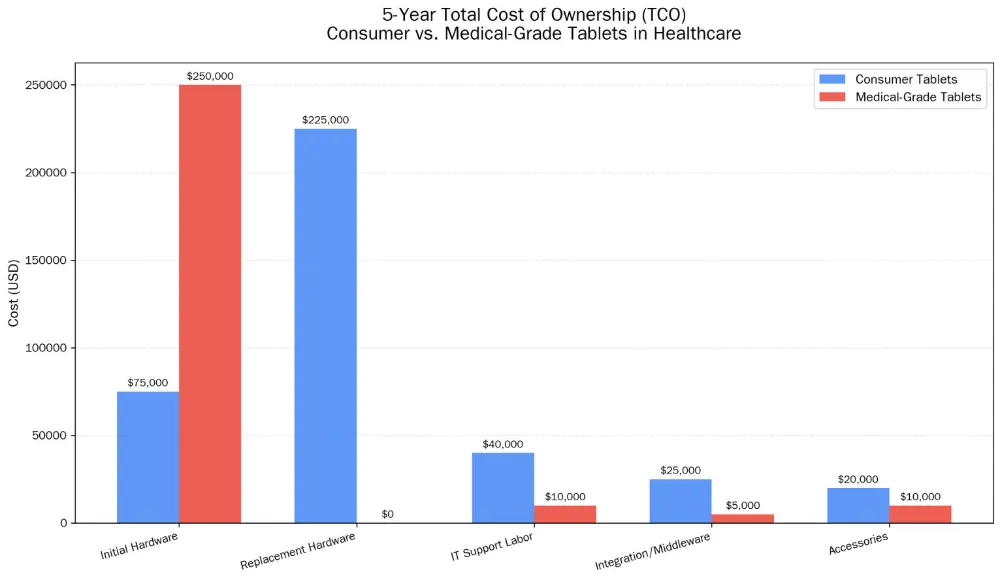
7.2 Total Cost of Ownership Calculation
When evaluating cost benefit analysis medical grade vs consumer tablets healthcare, total cost of ownership (TCO) over a 5-year period tells a different story:
| Cost Component | Consumer Tablets (5-year) | Medical-Grade Tablets (5-year) |
|---|---|---|
| Initial hardware | $75,000 | $250,000 |
| Replacement frequency | Every 18 months | Every 5 years |
| Replacement hardware cost | $225,000 (3 cycles) | $0 |
| IT support labor (failure management) | $40,000 | $10,000 |
| Integration/middleware | $25,000 | $5,000 |
| Accessories (cases, charging) | $20,000 | $10,000 |
| Total 5-year TCO | $385,000 | $275,000 |
Consumer tablets, despite lower acquisition cost, often exceed the total cost of medical-grade terminals over a typical replacement lifecycle due to higher failure rates, more frequent replacement, and additional infrastructure requirements.
7.3 Intangible Costs
Beyond direct financial costs, consumer tablet deployments incur intangible costs:
- Clinical inefficiency: Time lost to device failures and troubleshooting
- Regulatory risk: Potential citations from surveyors for non-compliant devices
- Infection risk: Increased HAI transmission vectors
- Staff frustration: Nursing dissatisfaction with unreliable technology
8. Use Case Scenarios: When Each Makes Sense
8.1 Clinical Patient-Facing Applications
For devices used by patients or at the bedside:
- Medical-grade tablets are required
- Patient safety requirements demand IEC 60601 compliance
- Infection control protocols require antimicrobial, cleanable surfaces
- Integration with nurse call and EHR is essential
8.2 Staff-Facing Clinical Applications
For devices used by clinicians at point of care:
- Medical-grade tablets are strongly recommended
- Durability requirements for high-use environments
- Barcode scanner integration for medication safety
- Hot-swappable batteries for continuous availability
8.3 Administrative and Non-Clinical Applications
For devices used in offices, break rooms, or administrative areas:
- Consumer tablets may be acceptable
- No patient contact reduces infection control requirements
- Regulatory compliance less stringent outside patient care areas
- Lower durability requirements for controlled environments
8.4 Can I Use Consumer Tablets in a Hospital Setting?
The question can I use consumer tablets in a hospital setting has a nuanced answer. Consumer tablets can be used in limited, non-clinical contexts: patient education in waiting rooms, administrative tasks in offices, or staff communications in break areas. However, for any use involving patient care, patient rooms, or the patient environment, medical-grade tablets are necessary to meet safety, compliance, and infection control requirements.

9. Implementation Considerations
9.1 Clinical Engineering Involvement
Clinical engineering departments should evaluate all devices entering patient care areas. Their assessment should include:
- IEC 60601 certification verification
- Electromagnetic interference testing in clinical environments
- Cleaning protocol validation
- Integration testing with existing clinical systems
9.2 IT Infrastructure Readiness
Deploying clinical tablets requires:
- Secure Wi-Fi infrastructure with clinical-grade QoS
- Mobile device management (MDM) for configuration and security
- Application packaging and deployment processes
- Help desk training for clinical device support
9.3 Nursing Engagement
Successful deployment requires nursing involvement from the beginning:
- Device placement and workflow integration
- Training and competency validation
- Feedback loops for continuous improvement
9.4 Cleaning Protocol Development
Establish cleaning protocols that:
- Specify approved cleaning agents and methods
- Define cleaning frequency
- Assign responsibility for cleaning
- Provide training for environmental services staff
10. Future Trends in Clinical Mobile Computing
10.1 Ambient Intelligence
Next-generation medical-grade terminals will incorporate ambient sensing to automatically capture clinical data and anticipate clinician needs.
10.2 5G Connectivity
5G networks will enable higher bandwidth for imaging, telemedicine, and real-time clinical collaboration.
10.3 AI-Powered Clinical Decision Support
Medical-grade tablets will increasingly incorporate AI capabilities for bedside clinical decision support, drug interaction checking, and predictive analytics.
10.4 Sustainability
Manufacturers are focusing on energy efficiency, recyclable materials, and reduced hazardous substances in compliance with evolving environmental standards.
Conclusion: Making the Right Choice
The decision between consumer and medical-grade tablets should be guided by use case, regulatory requirements, and total cost of ownership rather than acquisition price alone.
Choose medical-grade tablets when:
- Devices will be used in patient rooms or the patient environment
- Regulatory compliance (IEC 60601) is required
- Infection control is a priority
- Integration with clinical systems (EHR, nurse call) is needed
- Device durability and reliability are critical
- The deployment spans 3+ years
Consumer tablets may suffice when:
- Devices are used in administrative or non-clinical areas
- No patient contact occurs
- Integration with clinical systems is not required
- Short-term or pilot deployments with limited scale
For healthcare organizations, the question is not whether medical-grade tablets are worth the premium, but whether the risks and hidden costs of consumer devices justify the apparent savings. The evidence suggests that for clinical applications, the choice is clear: medical-grade terminals deliver superior safety, reliability, and total value over their lifecycle.
Frequently Asked Questions (FAQ)
Q1: Can I use an iPad in a patient room?
A: Using an iPad in a patient room is generally not recommended and may violate regulatory requirements. iPads and other consumer tablets lack IEC 60601 certification, which is the international standard for medical electrical equipment safety. Within the patient environment (typically within six feet of a patient), non-certified devices can pose electrical safety risks and may cause electromagnetic interference with life-sustaining equipment such as ventilators, infusion pumps, and cardiac monitors. For patient-facing applications, medical-grade tablets with appropriate certifications are required.
Q2: Why are medical-grade tablets so much more expensive than consumer tablets?
A: Medical-grade tablets cost 2 to 4 times more than consumer tablets due to several factors: regulatory compliance costs including IEC 60601 testing and certification; specialized materials such as antimicrobial housing and zero-pore surfaces that withstand hospital-grade cleaning; enhanced durability components designed for 30,000+ hour MTBF; clinical integration features including EHR and nurse call system compatibility; and extended warranties with clinical support infrastructure. When evaluated over a 5-year total cost of ownership, medical-grade tablets often prove more cost-effective due to lower replacement rates and reduced IT support burden.
Q3: Are consumer tablets HIPAA compliant?
A: Consumer tablets can be configured to meet HIPAA compliance requirements through proper security controls, encryption, and mobile device management. However, consumer tablets lack the built-in clinical security features of medical-grade devices, such as hardware-level encryption, single sign-on integration with hospital identity systems, and secure boot capabilities. More importantly, HIPAA compliance addresses only data security—it does not address patient safety requirements (IEC 60601), infection control standards, or clinical workflow integration that medical-grade tablets provide.
Q4: How do I clean a medical-grade tablet?
A: Medical-grade tablets are engineered to withstand hospital-grade disinfectants. Cleaning protocols typically specify approved agents such as quaternary ammonium compounds, hydrogen peroxide-based cleaners, or diluted bleach solutions. The zero-pore surfaces and sealed enclosures prevent fluid ingress during cleaning. Many medical-grade tablets are also UV-C disinfection compatible, allowing automated disinfection cycles. Always consult the manufacturer’s cleaning guidelines, as improper cleaning agents can damage the device and void the warranty.
Q5: What is the lifespan of a medical-grade tablet compared to a consumer tablet?
A: Medical-grade tablets typically provide 4 to 5 years of service life in clinical environments, with MTBF (Mean Time Between Failures) ratings of 30,000 to 50,000 hours. Consumer tablets deployed in clinical settings generally fail within 12 to 18 months due to cleaning chemical exposure, physical damage from drops, and battery degradation from continuous charging cycles. Medical-grade tablets also feature hot-swappable batteries that can be replaced without device downtime, extending usable life beyond the battery lifespan.
Q6: Do medical-grade tablets integrate with Epic, Cerner, or other EHR systems?
A: Yes, medical-grade tablets offer native integration with major EHR platforms including Epic, Cerner, Meditech, and others. Integration occurs through HL7 messaging protocols and FHIR APIs, enabling bidirectional data flow including patient identification, care team display, medication administration recording, and clinical documentation. Consumer tablets typically require middleware or custom application development to achieve similar integration, adding cost and complexity.
Q7: Can I use consumer tablets for medication administration verification?
A: Using consumer tablets for medication administration verification is not recommended. Medical-grade tablets support integrated barcode scanner integration that enables the “five rights” verification process (right patient, right medication, right dose, right route, right time) at the bedside. Consumer tablets lack this integrated capability and require separate scanners or manual entry, increasing the risk of medication errors and reducing workflow efficiency.
Q8: What IP rating should I look for in a hospital tablet?
A: The required IP rating depends on the clinical environment. For general clinical use, IP54 (dust-protected and splash-resistant) is the minimum standard. For environments with high fluid exposure—such as operating rooms, emergency departments, or behavioral health units—IP67 (dust-tight and temporary immersion protection) is recommended. IP69K (high-pressure, high-temperature washdown) is typically reserved for specialized environments such as sterile processing or certain laboratory settings.
Q9: Are there any circumstances where consumer tablets are acceptable in healthcare?
A: Consumer tablets may be acceptable in non-clinical applications where there is no patient contact, such as administrative offices, staff break rooms, waiting room information kiosks, or educational displays in non-patient areas. For any application involving patient care, patient rooms, or the patient environment, medical-grade tablets are necessary to meet safety, regulatory, and infection control requirements.
Q10: How do I justify the higher upfront cost of medical-grade tablets?
A: Justification should focus on total cost of ownership rather than acquisition cost. As demonstrated in Section 7, medical-grade tablets often cost less over a 5-year lifecycle when accounting for replacement frequency, IT support, integration costs, and accessories. Additionally, justification should include intangible benefits: regulatory compliance, infection prevention, staff productivity gains, and patient safety. For organizations subject to value-based care reimbursement, the impact on patient satisfaction and clinical outcomes provides additional financial rationale.
Latest Content
- The 2026 Definitive Guide to Patient Bedside Entertainment Terminals
- Beyond the Screen: The 2026 Playbook for Smart Bedside Infotainment Systems
- Integrated Bedside Infotainment: 2026 Implementation & Security Guide
- Revolutionizing Patient Care: A Comprehensive Guide to Hospital Television Systems
- Interoperability in Smart Wards:Why Integration Architecture Matters More Than Features

